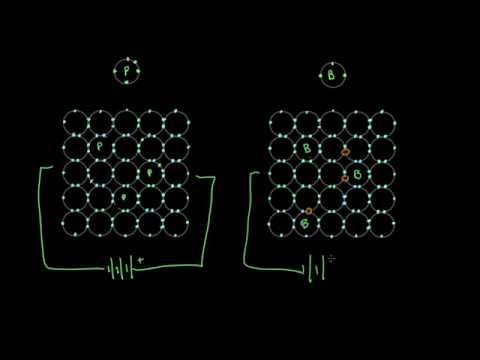
半導體的工作原理 (How semiconductors work)
 沒有此條件下的單字
沒有此條件下的單字US /məˈtɪriəl/
・
UK /məˈtɪəriəl/
- n. (c./u.)布料;素材;資料;材料;物質
- adj.重要的;物質的
US /ˈpɑzɪtɪv/
・
UK /ˈpɒzətɪv/
- adj.積極的;建設性的;確定的;正極的;積極的;有利的;陽性的;樂觀的;正數的;正像的
- n.正片
US /ˈnɛɡətɪv/
・
UK /'neɡətɪv/
- n.負電極的;否定詞;否定句;底片
- adj.消極的;負的;負面的;否定的;陰性的;負電的